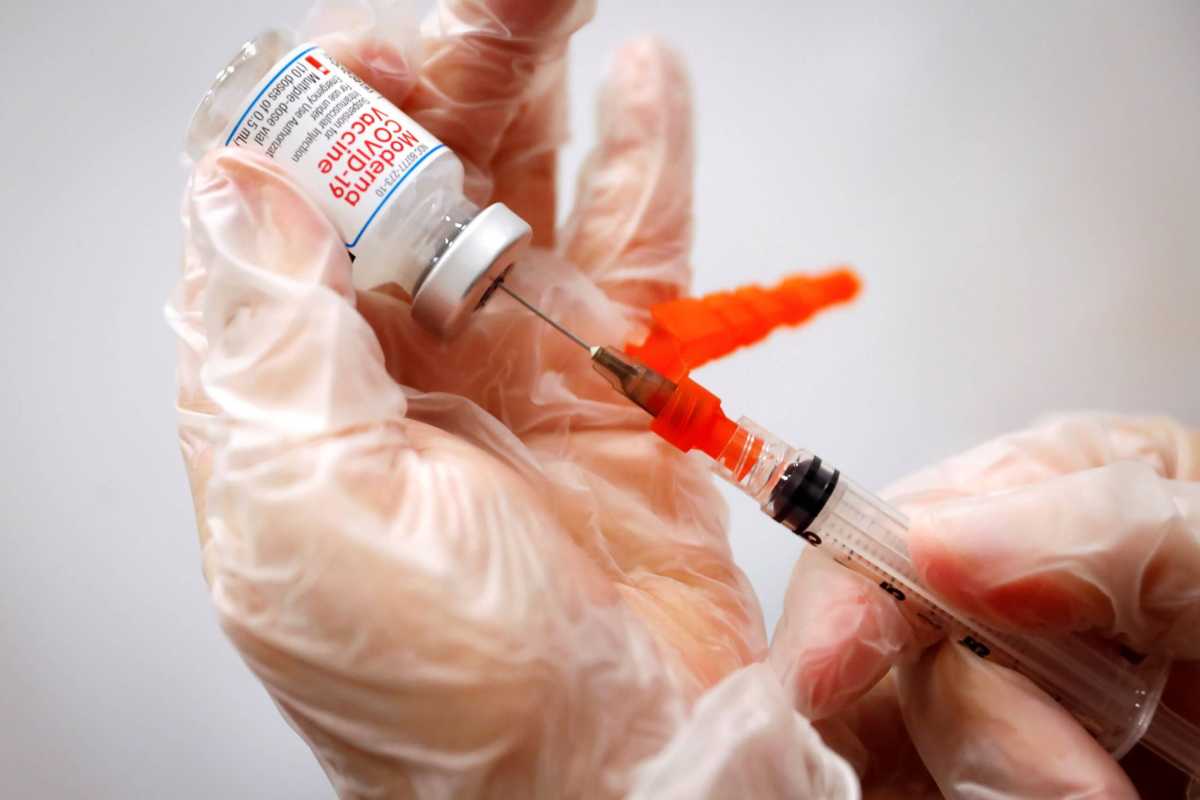
Following the utilization of mRNA technology in COVID-19 vaccines, Moderna is poised to launch phase one clinical trials for an mRNA HIV vaccine next month.
According to the National Institutes of Health’s (NIH) clinical trials registry, researchers are seeking approximately 56 participants between the ages of 18 to 50 years old to explore how mRNA technology protects adults against HIV. Researchers are providing four groups with two types of vaccines to test their effectiveness. Scientists believe the mRNA technology can “boost immunogens” that could aid the body’s response in blocking the virus. The clinical trials are slated to launch this fall and will continue until 2023.
Dr. Andrew Pekosz, a virologist and professor at John Hopkins School of Public Health, told Verywell that the mRNA technology could protect patients against more than just COVID-19.
“[mRNA] is a good platform to try against HIV,” Pekosz told Verywell. “But because the immunity that you need to protect from HIV is a little bit different from what you need from flu and COVID-19, it’s more important to do the large scale trials to really see how effective it could be.”
Doctors also say the technology used in the mRNA vaccine is a promising tool in fighting other public health issues.
“Based on its success in protecting against COVID-19, I am hopeful that mRNA technology will revolutionize our ability to develop vaccines against other pathogens, like HIV and influenza,” Dr. Rajesh Gandhi, an infectious diseases specialist at Massachusetts General Hospital and chair of the HIV Medicine Association, said in an interview with Verywell.
According to the trial’s guidelines, participants must be willing to undergo HIV testing and risk reduction counseling. Individuals assigned female and male at birth must use some form of birth control during the study. Participants with a history of excessive alcohol use, STIs, individuals living with HIV, engaging in sex work, or have had multiple sex partners within the last six months are excluded from the study.
Last month, researchers at the University of Oxford in the UK kicked off a similar clinical trial to test the effectiveness of a new HIV vaccine among people living with HIV and HIV-negative individuals.
Another trial — one for an HIV prevention shot given once every other month — ended early last year thanks to encouraging results among cisgender women. The shots, led by ViiV Healthcare, were found to be 89 percent more effective than Truvada.
To sign up for the Gay City News email newsletter, visit gaycitynews.com/newsletter.